Digital Technology in Healthcare Delivery: The DGOS publishes a roadmap for every actor in the system
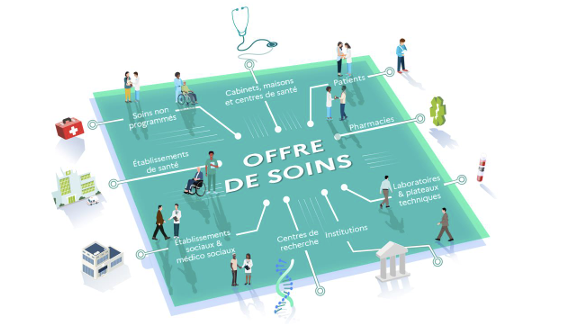
As healthcare professionnals’ practices continue to evolve, the General Directorate for Healthcare Provision (DGOS) has published a roadmap outlining the use of digital technology in healthcare delivery. They developed this initiative in collaboration with ministry officials, the National Health Insurance Fund (CNAM), regional health agencies (ARS), and industry stakeholders to highlight the benefits of using various digital tools.
Pharmacists and chronic conditions : Further expansion of their scope of practice

Published on November 28, 2024, and effective the following day, Decree No. 2024-1070 of November 26, 2024, further strengthens the authority of pharmacists regarding chronic conditions.
Teleconsultation companies: What changes under the Order of October 18, 2024

Since 2024, commercial companies have been able to offer telemedicine consultations provided by salaried medical professionals and covered by mandatory health insurance, provided they hold a license issued by the Ministry of Health.
Publication of a GDPR guide for compliance by microenterprises and SMEs by the European Data Protection Board

The European Data Protection Board (EDPB), an institution comprising all European data protection authorities, has published a practical guide for micro, small, and medium-sized enterprises (MSMEs).
Publication of new standard agreement templates : developements and implications

On August 3, 2024, updated versions of the standard form for human research were published by an order (‘arrêté’) dated July 29, 2024.
Access to and reuse of personal data online: The CNIL issues its recommandations

Le 12 juin 2024, à l’issue d’une consultation publique, la Commission Nationale de l’Informatique et des Libertés (CNIL) a publié ses recommandations sur l’ouverture (open data) et la réutilisation de données personnelles publiées sur Internet. Ces directives sont destinées à aider les entreprises à concilier leurs objectifs avec les droits des individus, tout en tirant parti des données accessibles en ligne.
AI Act: The compliance timeline is now underway !

À quelques jours de l’entrée en vigueur du Règlement sur l’intelligence artificielle, plus communément connu sous le nom d’AI ACT, l’heure est, pour les organisations, à la définition du calendrier de mise en conformité.
Cybersecurity in Healthcare Facilities: ANAP Releases an Action Plan

Healthcare providers are by far the most targeted group by cybercriminals: in 2022, 78% of cyberattacks occurred in the healthcare sector.
Regulations for healthcare facilities continue to take shape

On June 21, 2024, a decree was issued to specify the implementing rules for Law No. 2023-378 of May 19, 2023, regarding the regulation of health centers providing dental, ophthalmological, or orthoptic services, representing a total of more than 2,000 facilities.
